My lab focuses on cryo-electron tomography (cryo-ET) and its application in in-situ structural biology, especially in emerging enveloped viruses.
We aim at bridging dimensions between sub-nanometer resolution structures to micrometer scale in in-situ landscapes, a soaring demand in the field of structural biology. To achieve this, we are active in developing cryo-ET methods, including: 1) High throughput cryo-ET and subtomogram averaging at near-atomic resolution; 2) Focused-ion beam milling of cryo samples (cryo-FIB-SEM) and 3) Correlative light and electron microscopy (CLEM).
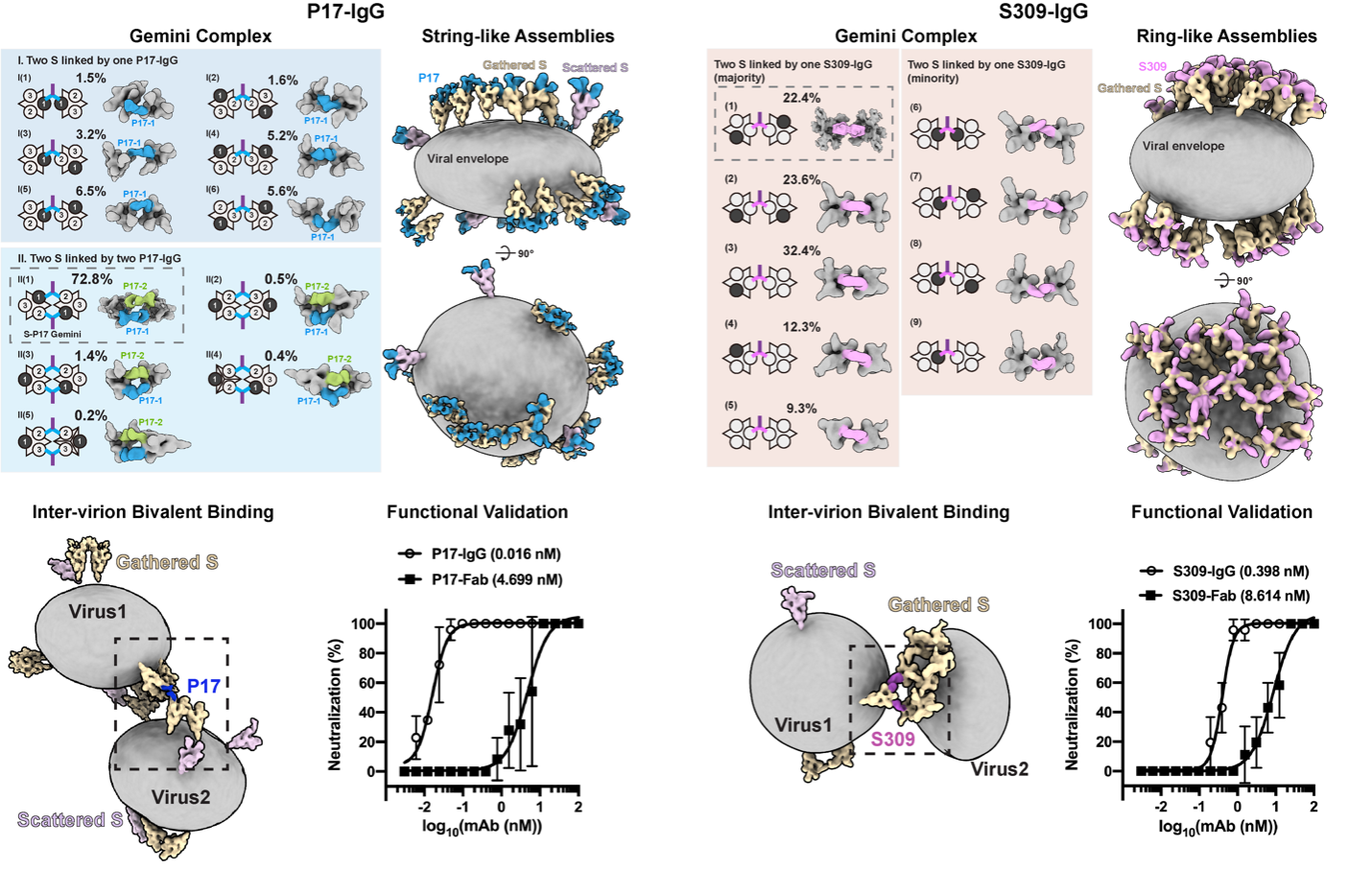
Among the vast applications, we primarily study the emerging enveloped viruses, which are the most threatening pathogens to the public health nowadays. Most enveloped viruses are pleomorphic and impose great challenge for structural biology, cryo-ET and subtomogram averaging are the primary methods in solving their native structures. We aim to shed light on: how are these viruses assembled, and how do they disassemble through membrane fusion, where are their weaknesses?